
Thus, the molar mass of bilirubin can be expressed as 584.73 g/mol, which is read as “five hundred eighty four point seventy three grams per mole. The division sign (/) implies “per,” and “1” is implied in the denominator. Now, compute the sum of products of number of atoms to the atomic weight: Molar mass (C 12 H 22 O 11 (sucrose)). 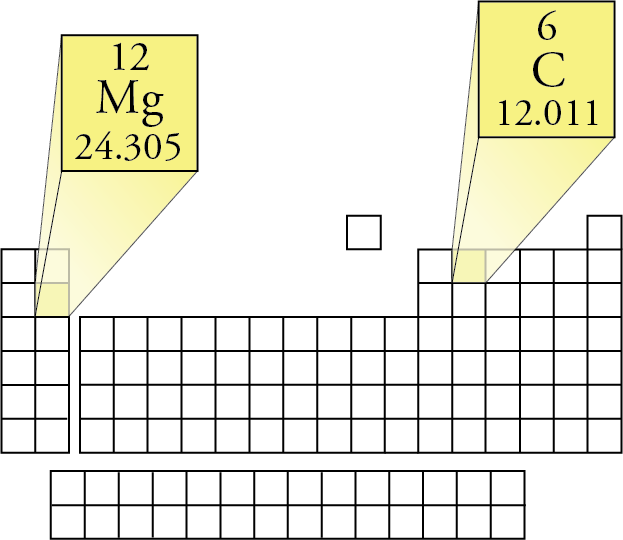
Then, lookup atomic weights for each element in periodic table: C: 12.0107, H: 1.00794, O: 15.9994. Referring to the periodic table, the atomic mass of K is 39.10 amu, and so its molar mass is 39.10 g/mol. /chemical-periodic-table-of-elements-with-color-cells-vector-illustration-840464844-ff61af51081b4fb1959504105e232e01.jpg)
For example, the molar mass of Ba(OH) 2 requires the sum of 1 mass of Ba, 2 masses of O, and 2 masses of H: The molar mass of Ba(OH)2 requires the sum of 1 mass of Ba, 2 masses of O, and 2 masses of H: 1 Ba molar mass:īecause molar mass is defined as the mass for 1 mol of a substance, we can refer to molar mass as grams per mole (g/mol). First, compute the number of each atom in C 12 H 22 O 11 (sucrose): C: 12, H: 22, O: 11. Because the definitions of both the mole and the atomic mass unit are based on the same reference substance, 12 C, the molar mass of any substance is numerically equivalent to its atomic or formula weight in amu. 1: Moles to Mass Conversion with Elements. According to the periodic table, 1 mol of U has a mass of 238.0 g, so the mass of 2 mol is twice that, or 476.0 g. For example, the atomic mass of carbon is 12.01 amu, and its molar mass is 12. The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g. Going back to 32 S, we know that because its molar mass is 31.97 grams per mole, its atomic mass must be 31.97 amu. The molar mass of an element is the same as its atomic mass, expressed in grams/mole. However, as you can see on the periodic table, sulfur is listed as 32.07 amunot 31.97 This is because the periodic table lists atomic weightsthe averages of the atomic mass for each one of the element’s stable isotopes. The molar mass is determined by adding up the atomic masses of all the atoms present in a molecule.The periodic table lists the molar masses of all the elements. In formulas with polyatomic ions in parentheses, the subscript outside the parentheses is applied to every atom inside the parentheses. Going back to 32 S, we know that because its molar mass is 31.97 grams per mole, its atomic mass must be 31.97 amu. By recognizing the relationship between the molar mass (g/mol), moles (mol), and particles, scientists can use dimensional analysis. For example, if the atomic mass of sulfer (S) is 32.066 amu, then its molar mass is 32.066 g/mol. As a result of this standard, the mass of all other elements on the periodic table are determined relative to carbon-12. Different molecules of the same compound may have different molecular masses because they contain different isotopes of an element. This is the standard unit for atomic or molecular mass, and 1 amu is thus 1/12 th the mass of a 12 C atom. adenosine (C 10H 13N 5O 4), a component of cell nuclei crucial for cell divisionīe careful when counting atoms. The molar mass of any element can be determined by finding the atomic mass of the element on the periodic table. The molecular mass (m) is the mass of a given molecule.The unit dalton (Da) is often used.The number 6. barium sulfate (BaSO 4), used to take X rays of the gastrointestional tract A mole is defined as exactly 6.02214076 × 10 23 particles, e.g., atoms, molecules, ions or electrons. One mole of a substance is equal to 6.022 × 10²³ units of that substance (such as atoms, molecules, or ions). 
What is the mass of 1 mol of each substance? \) it is the formula unit.\): Moles to Mass Conversion with Compounds When you open any file of an element in the periodic table, you will find a small table with some basic information about that element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
